 
Nevertheless, it can serve as a familiar reference point to orient yourself in a spectrum. This peak is not terribly useful, as just about every organic molecule that you will have occasion to analyze has these bonds. The jagged peak at approximately 2900-3000 cm -1 is characteristic of tetrahedral carbon-hydrogen bonds. Within that range, carboxylic acids, esters, ketones, and aldehydes tend to absorb in the shorter wavelength end (1700-1750 cm-1), while conjugated unsaturated ketones and amides tend to absorb on the longer wavelength end (1650-1700 cm -1). Notice how strong this peak is, relative to the others on the spectrum: a strong peak in the 1650-1750 cm -1 region is a dead giveaway for the presence of a carbonyl group. The key absorption peak in this spectrum is that from the carbonyl double bond, at 1716 cm -1 (corresponding to a wavelength of 5.86 mm, a frequency of 5.15 x 10 13 Hz, and a ΔE value of 4.91 kcal/mol). However, the utility of the fingerprint region is that the many bands there provide a fingerprint for a molecule. The fingerprint region is often the most complex and confusing region to interpret, and is usually the last section of a spectrum to be interpreted. Many different vibrations, including C-O, C-C and C-N single bond stretches, C-H bending vibrations, and some bands due to benzene rings are found in this region. This region is notable for the large number of infrared bands that are found there. The region of the infrared spectrum from 1200 to 700 cm -1 is called the fingerprint region. Group frequency and fingerprint regions of the mid-infrared spectrum These types of infrared bands are called group frequencies because they tell us about the presence or absence of specific functional groups in a sample.įigure 2. This makes these bands diagnostic markers for the presence of a functional group in a sample. For example, C-H stretching vibrations usually appear between 32cm -1 and carbonyl(C=O) stretching vibrations usually appear between 18cm -1. 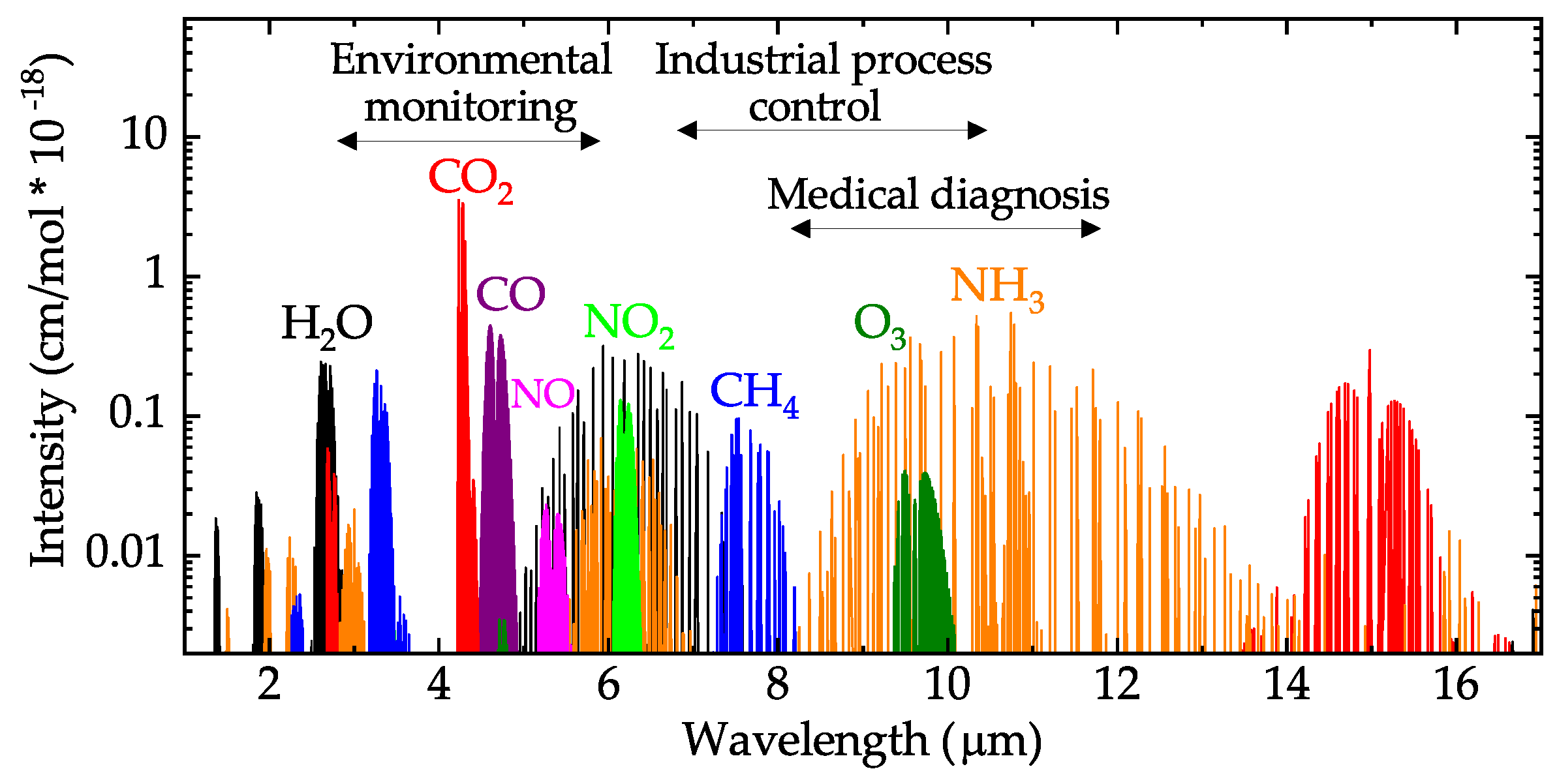
\)Īn important observation made by early researchers is that many functional group absorb infrared radiation at about the same wavenumber, regardless of the structure of the rest of the molecule. Provides a list of several representative examples.  
In one class of techniques there is a transfer of energy between the photon and the sample. We will divide spectroscopy into two broad classes of techniques. A spectroscopic measurement is possible only if the photon’s interaction with the sample leads to a change in one or more of these characteristic properties. In the previous section we defined several characteristic properties of electromagnetic radiation, including its energy, velocity, amplitude, frequency, phase angle, polarization, and direction of propagation. This change in wavelength as light passes between two media explains the refraction of electromagnetic radiation shown in Figure 10.1.1 If we replace the speed of light in a vacuum, c, with its speed in the medium, \(v\), then the wavelength is Because its velocity depends upon the medium in which it is traveling, the electromagnetic radiation’s wavelength, \(\lambda\), changes. When electromagnetic radiation moves between different media-for example, when it moves from air into water-its frequency, \(\nu\), remains constant. Wavenumbers frequently are used to characterize infrared radiation, with the units given in cm –1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
